In second grade, my oldest studied the layers of the ocean. As a fun activity at home, I thought we could bring what he learned alive by modeling the layers. As it turned out, making the layers of the ocean was also a great simple activity to teach density for kids!
Making the Layers of the Ocean-A Density Activity
3 Main Layers of the Ocean
The three main layers of the ocean are the surface ocean, the deep ocean, and the seafloor sediments. In our model of the ocean, we will use 3 substances (sand, salt water, and water) of different densities to create the ocean layers. We’ll also calculate the density of each substance.
What is Density?
Density is how much a substance weighs in a certain volume. For example, 1 cup of rocks weighs more than 1 cup of cheerios. So rocks have a higher density than cheerios. The formula for density is:
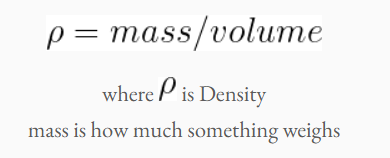
When in liquid, the material with the highest density always sinks to the bottom, and the lowest density material floats to the top. To create the deep ocean layer, we will increase he density of water by adding salt to it. Adding salt to water will increase the weight of the water, and the density too!
Supplies:
-water
-salt
-sand
–food coloring
–fish shaped ice tray
–1 cup measuring cup
–1/2 cup measuring cup
–digital scale
Procedure:
Step 1: Fill a cup with water, then add green food coloring to the water.
Step 2: Measure out 1/2 cup of the green water, and weigh it on the electronic scale. Write down the weight in table 1 below.
| Table 1: Finding the Density of Sand, Salt Water, and Water | |||
| Material | Weight | Volume | Density (oz/cup) |
| Water | |||
| Sand | |||
| Salt Water | |||
Step 3: Fill the fish shaped ice tray with the green colored water. Place the ice tray in the freezer.
Step 4: Fill the 1/2 cup measuring cup with sand.
Step 5: Weigh the measuring cup of sand, and write the weight down in table 1.
Step 6: Pour the sand into a clear, cylindrical cup or vase.
Step 7: Boil 6 cups of water, and add a cup of salt to the boiling water. Stir the hot water until all the salt is dissolved. Add a few drops of blue food coloring to the salt water, and stir. Set aside to cool.
Step 8: Once the blue water from step 6 is room temperature, measure 1/2 cup of the salt water, and weigh it on the electronic scale. Write the weight down in table 1.
Step 9: Pour 1 cup of the blue salt water from steps 7 and 8 into the vase from step 6.
Step 10: Take the ice tray out of the freezer. Place 6 ice fish into the cup of sand and salt water.
As the ice melts, the green water remains as the top layer, and the sand stays at the bottom of the vase.
What Happened?
Let’s start by looking at Table 1 with all the data filled in:
| Table 1: Finding the Density of Sand, Salt Water, and Water | |||
| Material | Weight (oz) | Volume (cup) | Density (oz/cup) |
| Water | 5.25 | 1/2 | 10.5 |
| Sand | 6.8 | 1/2 | 13.6 |
| Salt Water | 5.8 | 1/2 | 11.6 |
Which material has the highest density? The sand! The next highest is the salt water, and the water has the lowest density. Remember that the highest density always sinks to the bottom, and the lowest density material floats to the top? In the ocean, this is exactly what happens too. The material with the highest density, the seafloor sediments, settle to the bottom of the ocean. The deep ocean has a higher density than the surface ocean because it is significantly cooler than the surface ocean, so it settles in between the seafloor sediments and the surface ocean. The surface ocean has the lowest density because of its higher temperature from being exposed to sunlight.
What other ways can you think of to explore density? I would love to learn of your ideas!
